A) 1 half-life
B) 2 half-lives
C) 3 half-lives
D) 4 half-lives
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The first-order reaction,2 N2O(g) → 2 N2(g) + O2(g) ,has a rate constant equal to 0.76 s-1 at 1000 K.How long will it take for the concentration of N2O to decrease to 42% of its initial concentration?
A) 0) 88 s
B) 1) 1 s
C) 1) 8 s
D) 2) 4 s
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a reaction that follows the general rate law,Rate = k[A][B]2,what will happen to the rate of reaction if the concentration of B is increased by a factor of 3.00? The rate will
A) decrease by a factor of 1/ 9.00.
B) decrease by a factor of 1/ 3.00.
C) increase by a factor of 3.00.
D) increase by a factor of 9.00.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is true for the general rate law: Rate = k[A]m[B]n?
A) It can be written from the stoichiometry of the overall reaction.
B) The overall order of the reaction is equal to m times n.
C) The values for the exponents must be determined by experiment.
D) The exponents in the rate law must be positive integers.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the first-order decomposition of A molecules (shaded spheres) in two vessels of equal volume.How will the half-life of decomposition in vessel (a) be affected if the volume of the vessel is decreased by a factor of 2? 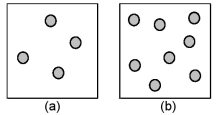
A) decrease by 1/2
B) increase by 2
C) increase by 4
D) stay the same
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which term describes the measure of the increase in the concentration of a product per unit time?
A) activation energy
B) kinetics
C) reaction rate
D) reaction time
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following figures represents the decay of 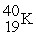 By positron emission to give
By positron emission to give 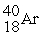 After three half-lives has passed? Unshaded spheres represent
After three half-lives has passed? Unshaded spheres represent 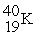 Atoms and shaded spheres represent
Atoms and shaded spheres represent 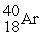 Atoms.
Atoms. 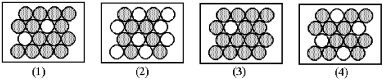
A) figure (1)
B) figure (2)
C) figure (3)
D) figure (4)
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the first-order reaction,2 N2O(g) → 2 N2(g) + O2(g) ,what is the concentration of N2O after 3 half-lives if 0.15 mol of N2O is initially placed into in a 1.00-L reaction vessel?
A) 9) 4 × 10-3 M
B) 1) 9 × 10-2 M
C) 3) 8 × 10-2 M
D) 7) 5 × 10-2 M
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Chlorine reacts with chloroform according to the reaction given below: Cl2 + CHCl3 → CCl4 + HCl When the initial concentration of Cl2 is doubled the reaction rate increases by a factor of 1.41.What is the order of the reaction with respect to Cl2?
A) -1/2
B) -1
C) 1/2
D) 2
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The decomposition of ozone in the stratosphere can occur by the following two-step mechanism: Step 1: Br + O3 → BrO + O2 Step 2: BrO + O → Br + O2 Which species is an intermediate in this mechanism?
A) Br
B) BrO
C) O
D) O3
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
The decomposition of hydrogen peroxide occurs according to the equation 2 H2O2(aq)→ 2 H2O(l)+ O2(g) A concentration-time study of this reaction produces a straight line when ln[H2O2] is plotted versus time.Therefore,this is a ________ order reaction.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What fraction of collisions will have sufficient energy to react for a gas whose activation energy is 68 kJ/mol at 25°C?
A) 1) 2 × 10-12
B) 2) 7 × 10-2
C) 0) 96
D) 8) 3 × 1011
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydroquinone,HOC6H6OH,can be formed by the reaction with acetylene below:
2 HCCH + 3 CO + H2O → HOC6H4OH + CO2
How is the rate of disappearance of acetylene,HCCH,related to the appearance of hydroquinone (Hq) ? 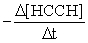 = ?
= ?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isomerization reaction,CH3NC → CH3CN,is first order and the rate constant is equal to 0) 46 s-1 at 600 K.What is the concentration of CH3NC after 0.20 minutes if the initial concentration is 0.30 M?
A) 1) 2 × 10- 3 M
B) 2) 7 × 10- 3 M
C) 1) 2 × 10- 1 M
D) 2) 7 × 10- 1 M
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following does not affect the rate of a bimolecular reaction?
A) concentrations of reactants
B) presence of a catalyst
C) temperature
D) All of these affect the rate.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a particular first-order reaction,it takes 24 minutes for the concentration of the reactant to decrease to 25% of its initial value.What is the value for rate constant (in s-1) for the reaction?
A) 2) 0 × 10- 4 s-1
B) 9) 6 × 10- 4 s-1
C) 1) 2 × 10- 2 s-1
D) 5) 8 × 10- 2 s-1
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction: 2 HI → H2 + I2,is second order and the rate constant at 800 K is 9.70 × 10-2 M-1 s-1.How long will it take for 8.00 × 10-2 mol/L of HI to decrease to one-fourth of its initial concentration?
A) 0) 619 s
B) 124 s
C) 387 s
D) 429 s
F) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
At an elevated temperature the decomposition of a gaseous oxide,AO2 occurs with a rate constant, k = 0.54 M-1s-1.If the half-life of this reaction is 926 seconds when [AO2] = 2.0 × 10-3 M and 462 seconds when [AO2] = 4.0 × 10-3 M,this reaction is ________ order.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider a reaction that occurs by the following one-step mechanism:
A2 + B2 → 2 AB
The potential energy profile for this reaction is shown below.  -The activation energy for the forward reaction is given by the difference in energy between which two reaction stages?
-The activation energy for the forward reaction is given by the difference in energy between which two reaction stages?
A) reaction stage 2 - reaction stage 1
B) reaction stage 2 - reaction stage 3
C) reaction stage 1 - reaction stage 3
D) reaction stage 3 - reaction stage 1
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction between chlorine and nitric oxide to form nitrosyl chloride is shown below.If the reaction rate doubles when the concentration of Cl2 is doubled and the rate quadruples when the concentration of NO is doubled,by what factor will the rate increase if both concentrations,NO and Cl2,are doubled? Cl2(g) + 2 NO(g) → 2 NOCl(g)
A) 2
B) 4
C) 8
D) 16
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 181 - 200 of 206
Related Exams